umj Sulfur is an undesired element found in crude oil in form of sulfur compound called mercaptan. Once crude oil go through various process unit, the sulfur get removed from the end product but it remain present in form of H2S in different streams. This waste gas cannot be released in open atmosphere because of adverse impact H2S on human and environment. Hence, to destroy H2S and capture sulfur , Sulfur recovery unit is required in crude oil refinery. Government do not release clearance letter to any new crude oil refinery project until project have Sulfur recovery unit in their plan.
Crude Oil Industry process units can be categorized in three categories:-
- Distillation/ Separation Unit.
- Product purification/Modification Unit.
- Process unit to meet environment norms.
- Utilities and offsites.
Sulfur recovery unit comes under 3rd category. As sulfur is available in abundance , it is always regarded as byproduct of crude oil refinery.
Chemical process of Sulfur recovery unit
After passing through various process units, sufur from mercaptan get removed and remain available in form of H2S. So, available standard chemical process to covert H2S to Sulfur is “Claus process”.
Chemical reactions can be tabulated as below:-
- 2 H2S + 3 O2 → 2 SO2 + 2 H2O (ΔH = -518 kJ mol−1)
-
- 2 H2S + SO2 → 3 S + 2 H2O (This is called Claus Reaction)
- Combination of above two equations can be written as
-
- 2 H2S + O2 → 2 S + 2 H2O
A typical process flow diagram is as below-
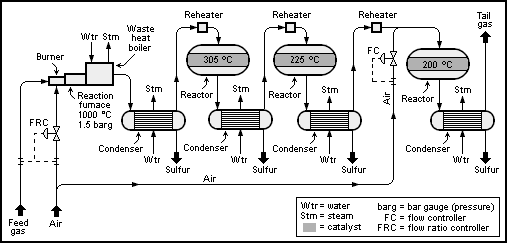
We will elaborate process details and construction details in next posting.
For Sulfur recovery Unit :-
Feed :-
- Acid Gas (H2S and CO2)
- Sour Gas (H2S and NH3)
Product:-
- Sulfur
Uses of Sulfur:-
- For H2SO4 (Sulfuric acid manufacturing)
- Medicine
- Cosmetics
- Fertilizer
- Rubber
- Pesticides
Sulfur market price is 270 USD/MT as on Dec 2021.



